Refractometer Uses in Industry and Selection
By Angelo DePalma, PHD
Refractometer uses: Characterizes fluids based on their refractive index –the refraction occurs due to the change in the speed of light as it travels through a material of one density to one of different density. By definition a vacuum has a refractive index of 1, and the refractive index of a solution or pure fluid is calculated by the equation:
Refractive Index = speed of light in a vacuum/speed of light through the substance
Light passes more slowly through media as the samples become denser or more concentrated. Hence, air has a refractive index of 1.000293, water’s Refractive Index is 1.33298 at 20C, and benzene’s is 1.501, while the Refractive Index of the anesthetic Sevoflurane ranges from 1.2745 to 1.2760.
Refractive Index is based on Snell’s Law, which describes the relationship between the angle of incidence and the angle of diffraction for light hitting the boundary between two different media, for example a liquid solution and air. Wikipedia provides a good background on Refractive Index and Snell’s Law.
Anyone who has seen the oar of a rowboat “bend” as it entered the water has experienced refraction first-hand. The phenomenon arises from the slower propagation of light waves through water compared with air. Commercial refractometers work on the same principle, although the degree of “bending” is measured precisely.
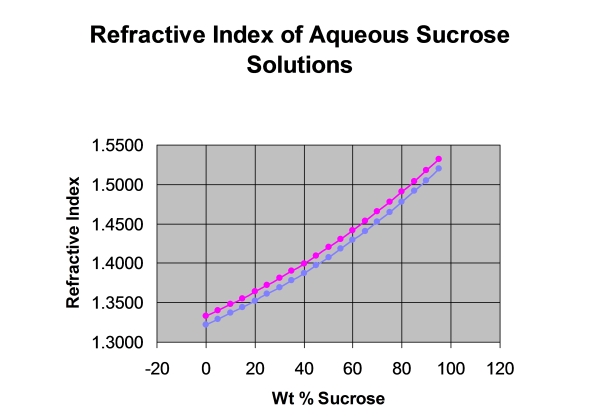
Refractive Index is a unique, constant physical characteristic of pure substances, such as lemon oil, but its most common use is for calculating the concentration of dissolved substances in water. Measuring the sugar content in water (“brix”) is a common application. Figure 1 illustrates that the higher the concentration, the higher the Refractive Index .
Refractometer Uses – Wavelength and Temperature
Refractometry measurements strongly depend on the wavelength of light applied to the sample, and the sample temperature. Standard refractometers use the wavelength 589.3 nanometers, (omit-the emission from the sodium D line), and take measurements at 20º C. While the wavelength is fixed, sample temperatures will vary unless controlled, and significantly affect refractometry measurements.
Liquids and solutions become less dense at higher temperatures, so light passing through higher-temperature samples encounter fewer solute (and solvent) molecules. One would therefore expect the Refractive Index at 25º C to be slightly lower than at 20º C. One way to compensate for temperature is to apply a numeric correction for temperature variations from 20º C –
Reasonable numeric correction based on ICUMSA correction tables work well for pure sugar and water substances where there is a linear relationship between Refractive Index and temperature change. The further one’s dissolved substance becomes less pure and the further the sample is from 20C the greater the potential mathematical error. The most common temperature correction formula applied is the one developed by ICUMSA when they created the BRIX table that approximates the difference between the Refractive Index at measurement and at reference temperatures.
Refractive Index Correction factors differ for all solutions and pure liquids. Some high-end instruments apply a correction factor automatically, a feature known as automated temperature compensation or automated temperature correction. The most reliable solution to non-standard temperatures, however, is the ability to heat or cool the sample to precisely 20º C. The most efficient systems provide heating and cooling from both above and below the sample, and employ Peltier heating/cooling rather than the traditional water bath.
Water bath maintenance is a time-consuming exercise involving changing and topping off the water periodically, and adding algaecide. Plus the temperature of the water bath itself requires measurement and calibration. Electronic Peltier temperature control is more compact than water baths and provides greater temperature accuracy and more rapid equilibration.
Unlike spectroscopy, which can identify substances uniquely based on their absorption, Refractometry is useful only when the material being analyzed is known. For example a salt solution at one concentration may have the same Refractive Index as a sugar solution of a different concentration. Despite this limitation, refractometers are ubiquitous in industrial and scientific laboratories, particularly in quality control settings where a rapid Refractive Index measurement indicates if the product is on specification.) The following applications are just the tip of the iceberg of possibilities:
- Salinity of water
- Sweetness of beverages
- Hydrocarbon content of motor fuels
- Brix, or sugar concentration in a fermentation
- Concentration of maple syrup
- Protein in solution
Refractometer Uses and Selection for Industry
Because laboratories have widely differing needs based on their applications, measurement environment, compliance requirements, operator training and experience level refractometer vendors have responded by offering numerous models to meet the various industry settings. These range from desktop units to handheld, electronic/digital to manual, with varying levels of precision and documentation.
For every-day users, performance is an obvious factor in purchasing a refractometer. Individuals who buy strictly on price may be missing on value. Refractometers tend to last a long time if properly maintained. Realizing the full worth of an analytical instrument requires that the purchaser understand the value of their measurements, frequency of use, and the cost or lost opportunity of an incorrect reading. The difference in cost between a low-end instrument and a quality refractometer may amount to just a few cents per measurement over the instrument’s lifetime.
Small, handheld or palm-sized refractometers costing between $200 and $2,000 are generally used for specific tasks like testing wine or antifreeze. These instruments may be electronic or battery-powered, or non-electronic models that rely on ambient light and require the user to make a visual determination. The electronic/digital designs tend to be somewhat larger and more expensive, but they provide greater accuracy. Small refractometers are ideal for field work, home, for bringing the “lab” to the sample instead of the other way around.
Laboratory Refractometers in the $7,000 to $14,000 price range are workhorse instruments for large manufacturing companies seeking to maintain a high level of quality and adherence to stringent quality guidelines. The higher cost is justified by the instruments’ superior optics and performance. Where handheld models provide resolution between 0.1 and 0.01, lab instruments get down to 0.0001. These refractometers typically interface with a computer and are the instruments of choice for high-throughput labs seeking both accuracy and data integrity.
For research applications or those requiring very high accuracy, top-of-the-line laboratory refractometers provide accuracy to 0.00001 and full Refractive Index measurement range, up to 1.70000. These instruments feature connectivity to computers as well as other instrumentation.
In addition to these three laboratory refractometer categories, companies working in process industries might consider in-line refractometers that measure Refractive Index continuously.
Conclusion
Refractometry is a time-proven analytical method that remains a staple of modern laboratories. Selecting the right refractometer depends on your need for accuracy, sample throughput, location of the measurement, and interoperability with other instruments, and requirements for data handling and processing.


 Download a PDF
Download a PDF